In an earlier post on this topic,[cite]10.59350/f00wf-5tq46[/cite] ‡ I described how the curly-arrows describing the mechanism of a nucleophilic addition at a carbonyl group choreograph in two distinct ways, as seen in red or blue below.

Respiratory pigments are metalloproteins that transport O 2 , the best known being the bright red/crimson coloured hemoglobin in human blood. The colour derives from Fe 2+ at the core of a tetraporphyrin ring. But less well known is blue blood , and here the colour derives from an oxyhemocyanin unit based on Cu 1+ (the de-oxy form is colourless) rather than iron.
One of the important aspects of chemical reaction mechanisms is the order in which things happen. More specifically, the order in which bonds make or break when there are more than two involved in undertaking a reaction.
What’s a Journal For? This debate has been raging ever since preprint servers were introduced as far back as 1991!
Sometimes you come across a reaction which is so simple in concept that you wonder why it took so long to be accomplished in practice.
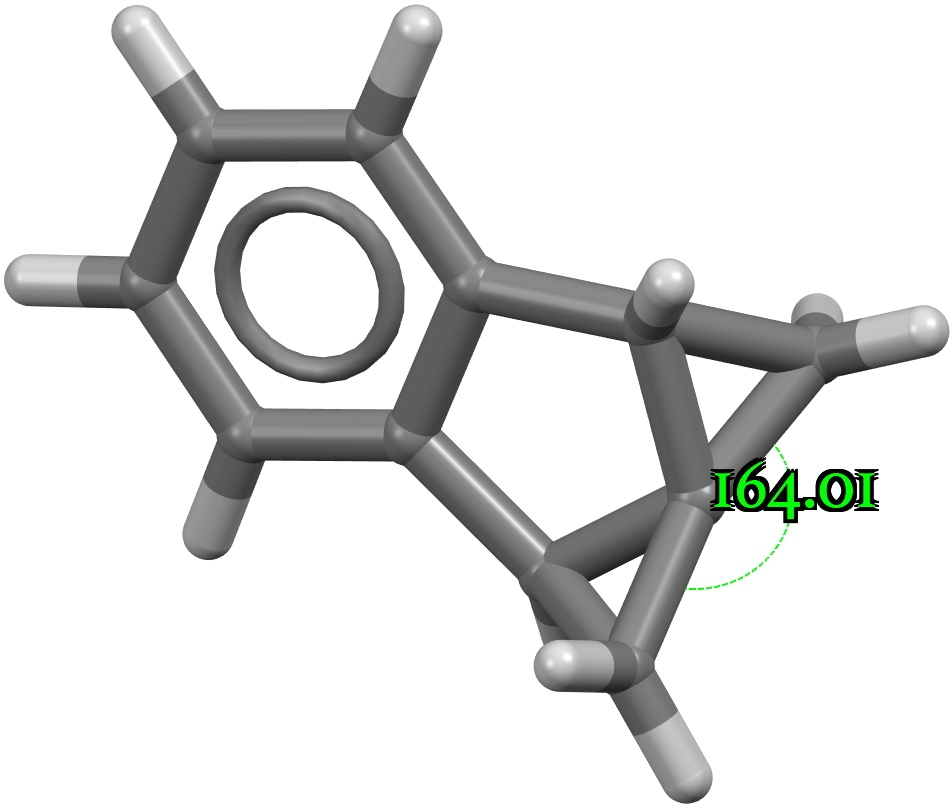
Four-coordinate carbon normally adopts a tetrahedral shape, where the four angles at the carbon are all 109.47°. But how large can that angle get, and can it even get to be 180°? A search of the CSD (crystal structure database) reveals a spiropentane as having the largest such angle, VAJHAP with 164°[cite]10.1021/ja00186a058[/cite] Because crystal structures might have artefacts such as disorder etc, it is always good to check this with a
Previously, a mechanism with a reasonable predicted energy was modelled for the isomerisation of an oxetane carboxylic acid to a lactone by using two further molecules of acid to transfer the proton and in the process encouraging an Sn2 reaction with inversion to open the oxetane ring. We are now ready to explore variations to this mechanism to see what happens.
I have long been fascinated by polymers of either carbon dioxide, † or carbon monoxide, or combinations of both. One such molecule, referred to as dioxane tetraketone when it was featured on the ACS molecule-of-the-week site and also known as the anhydride of oxalic acid, or more formally 1,4-dioxane-2,3,5,6-tetraone, has been speculated upon for more than a century.[cite]10.1002/cber.19080410335[/cite] The history of
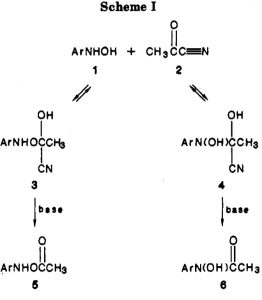
Minds (and memories) can work in wonderful ways. In 1987[cite]10.1021/jo00389a050[/cite] we were looking at the properties of “stable” tetrahedral intermediates formed in carbonyl group reactions. The reaction involved adding phenylhydroxylamine to acetyl cyanide.
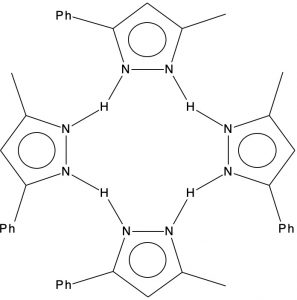
Previously, I explored the unusual structure of a molecule with a hydrogen bonded interaction between a phenol and a pyridine. The crystal structure name was RAKQOJ and it had been reported as having almost symmetrical N…H…O hydrogen bonds. This feature had been determined using neutron diffraction crystallography, which is thought very reliable at determining proton positions.
