Recollect this earlier post on the topic of the Baeyer-Villiger reaction. In 1999 natural abundance kinetic isotope effects were reported[cite]10.1021/ja992016z[/cite] and I set out to calculate the values predicted for a particular model constructed using Quantum mechanics. This comparison of measurement and calculation is nowadays a standard verification of both experiment and theory.
Messages de Rogue Scholar
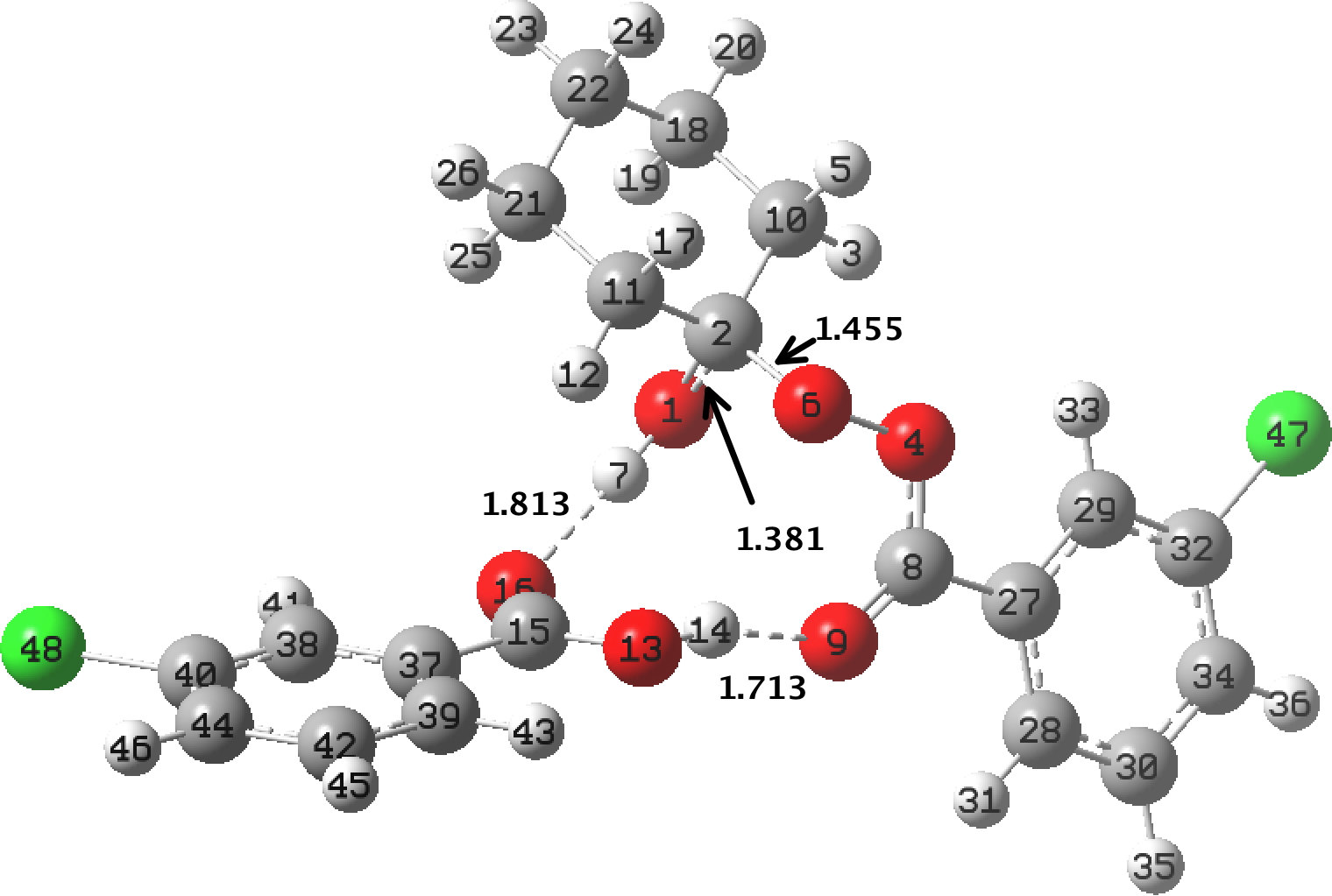
In the preceding post, I discussed the reaction between mCPBA (meta-chloroperbenzoic acid) and cyclohexanone, resulting in Baeyer-Villiger oxidation via a tetrahedral intermediate (TI). Dan Singleton, in whose group the original KIE (kinetic isotope measurements) were made, has kindly pointed out on this blog that his was a mixed-phase reaction, and that mechanistic comparison with homogenous solutions may not be
I have blogged before about the mechanism of this classical oxidation reaction. Here I further explore computed models, and whether they match the observed kinetic isotope effects (KIE) obtained using the natural-abundance method described in the previous post. There is much previous study of this rearrangement, and the issue can be reduced to deciding whether TS1 or TS2 is rate-limiting.
My PhD thesis involved determining kinetic isotope effects (KIE) for aromatic electrophilic substitution reactions in an effort to learn more about the nature of the transition states involved.[cite]10.1039/p29750001209[/cite] I learnt relatively little, mostly because a transition state geometry is defined by 3N-6 variables (N = number of atoms) and its force constants by even more and you get only one or two measured KIE per reaction;
Allotropes are differing structural forms of the elements. The best known example is that of carbon, which comes as diamond and graphite, along with the relatively recently discovered fullerenes and now graphenes. Here I ponder whether any of the halogens can have allotropes. Firstly, I am not aware of much discussion on the topic.
Sodium borohydride is the tamer cousin of lithium aluminium hydride (LAH). It is used in aqueous solution to e.g. reduce aldehydes and ketones, but it leaves acids, amides and esters alone. Here I start an exploration of why it is such a different reducing agent. Initially, I am using Li, not Na (X=Li), to enable a more or less equal comparison with LAH, with water molecules to solvate rather than ether (n=2,3,5) and R set to Me.
Previously on this blog: modelling the reduction of cinnamaldehyde using one molecule of lithal shows easy reduction of the carbonyl but a high barrier at the next stage, the reduction of the double bond.
The reduction of cinnamaldehyde by lithium aluminium hydride (LAH) was reported in a classic series of experiments[cite]10.1021/ja01197a060[/cite],[cite]10.1021/ja01202a082[/cite],[cite]10.1021/ja01190a082[/cite] dating from 1947-8. The reaction was first introduced into the organic chemistry laboratories here at Imperial College decades ago, vanished for a short period, and has recently been reintroduced again. ‡ The experiment is
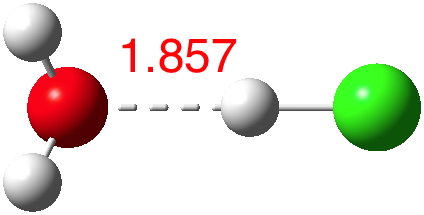
According to Guggemos, Slavicek and Kresin, about 5-6![cite]10.1103/PhysRevLett.114.043401[/cite]. This is one of those simple ideas, which is probably quite tough to do experimentally. It involved blasting water vapour through a pinhole, adding HCl and measuring the dipole-moment induced deflection by an electric field.
The journal of chemical education has many little gems providing inspiration for laboratory experiments. Jonathan Piard reports one based on the reaction below[cite]10.1021/ed4005003[/cite]; here I investigate the mechanism of this transformation. There are two things going on here;