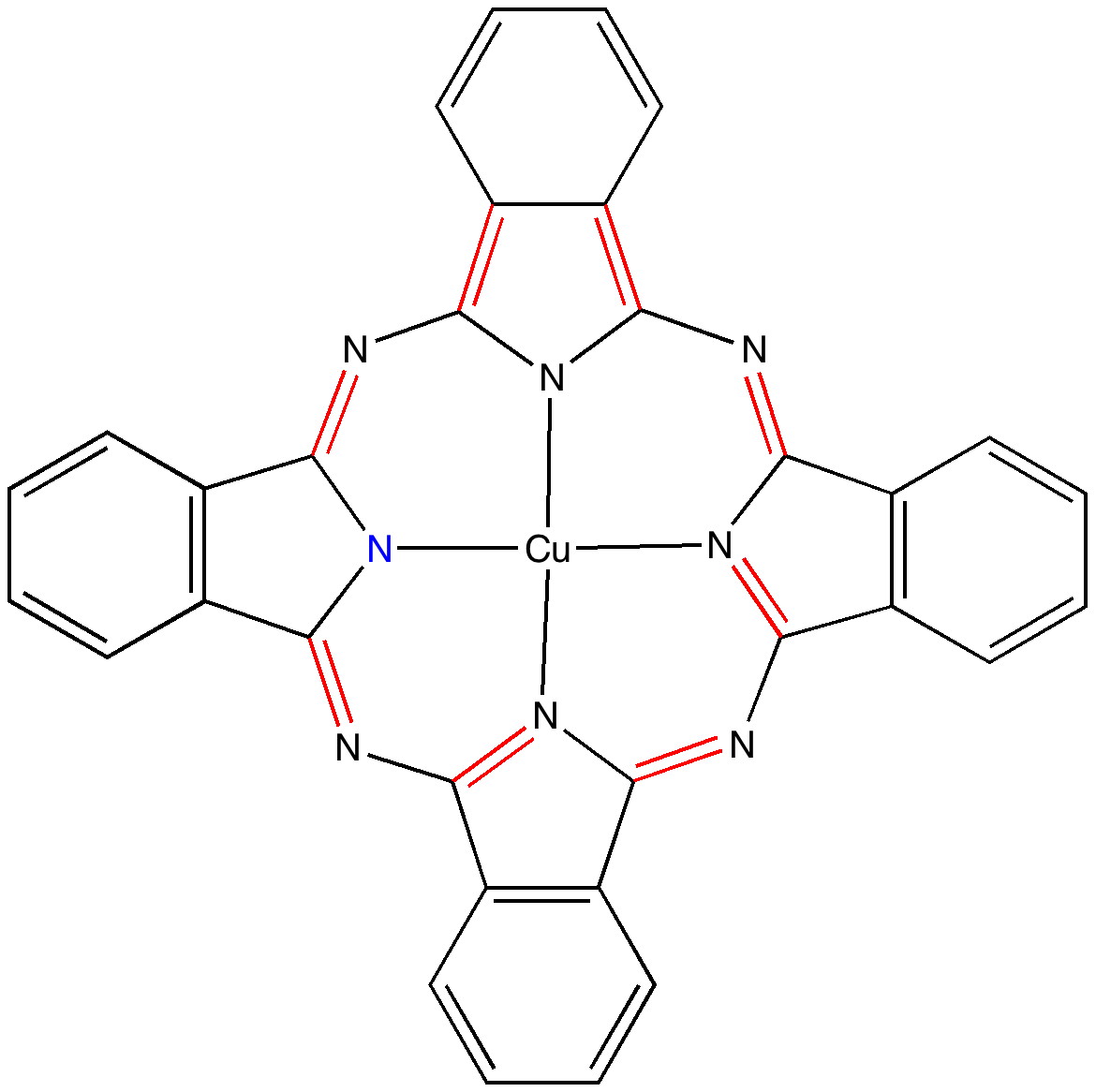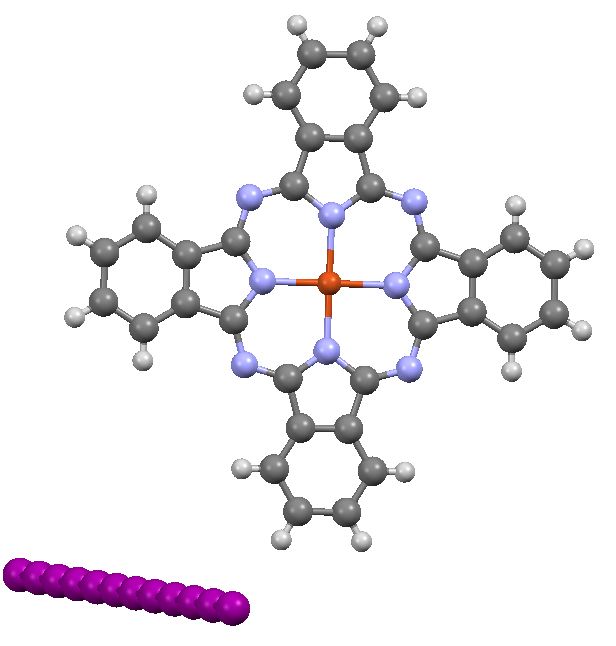
In the previous post I pondered the colour of Monastral blue (copper phthalocyanine). Something did not quite fit, and so I speculated that perhaps some oxidation of the pigment might give a new species. This species (Cambridge code FEGJOQ) comprises two parts of copper phthalocyanine, 1 part of the corresponding cation, and 1 part of triodide anion.