Au and Pt π-complexes of cyclobutadiene.
In the preceding post, I introduced Dewar's π-complex theory for alkene-metal compounds, outlining the molecular orbital analysis he presented, in which the filled π-MO of the alkene donates into a Ag+ empty metal orbital and back-donation occurs from a filled metal orbital into the alkene π* MO. Here I play a little "what if" game with this scenario to see what one can learn from doing so.
Firstly, I will use Au+ instead of Ag+, so as to make a comparison with Pt2+ a little more direct. The electronic configurations are of course [Xe].4f14.5d10.6s0 and [Xe].4f14.5d8.6s0 respectively. I will also replace a simple ethene with cyclobutadiene, the intent here being that this cyclo-diene is a very much better π-donor due to its anti-aromatic character. It also now has the possibility of acting as a four or a two-electron donor. I started with M=Pt+[cite]10.6084/m9.figshare.703546[/cite] by adding another double bond to the structure of the ethene complex.
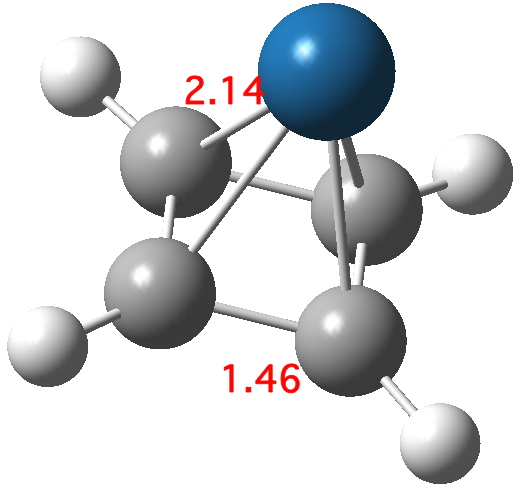
Optimising this starting structure in fact moves the metal and the final geometry has C4v symmetry; in other words the metal is bound symmetrically to all four carbons. The four C-C lengths are all the same (1.46Å) and strongly suggest that four electrons from the cyclobutadiene are participating in bonding; the Pt2+ is clearly capable of accepting four electrons, two into 6s0 and two into 5d8. In the process, the cyclobutadiene looses its antiaromaticity. The molecular orbitals of this species are all lovely; I illustrate just one below.
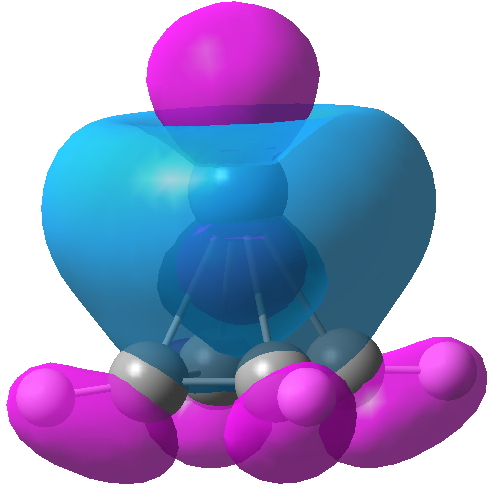
Click for 3D.
If the Pt in this C4v structure is mutated into Au+, the resulting optimised stationary point exhibits a negative force constant characteristic of a transition state[cite]10.6084/m9.figshare.703547[/cite]. As the d-shell is already fully, the Au can only accept two electrons, and this is therefore a nice illustration of the "18-electron" rule in operation. So, the Au+ complex must exist in at least one lower energy form. For example, one where the Au+ is coordinated to only one alkene is 94 kcal/mol lower in free energy.[cite]10.6084/m9.figshare.703576[/cite] This form results in electrons from the coordinated alkene being donated into the 6s Au orbital, and this action reduces the anti-aromaticity of the cyclobutadiene ring.
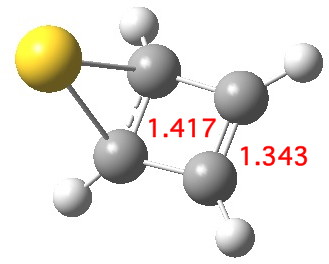
Another isomer also achieves this result, resulting in a further
lowering in free energy of 11.0
kcal/mol[cite]10.6084/m9.figshare.703577[/cite] The anti-aromaticity
this time is eliminated by forming an allyl cation on the ring. I have
described this mode in another post, commenting on the effect when a
guanidinium cation interacts with cyclobutadiene.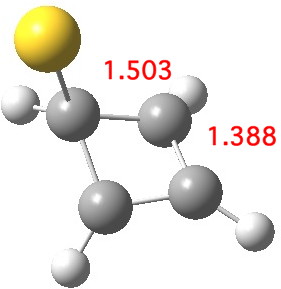
We have learnt that cyclobutadiene has many modes for eliminating 4n-electron antiaromaticity and other destabilising influences upon the ring. It can accept four electrons from a suitable acceptor (Pt2+), or two electrons from Au+ in two different ways.
Additional details
Description
In the preceding post, I introduced Dewar's π-complex theory for alkene-metal compounds, outlining the molecular orbital analysis he presented, in which the filled π-MO of the alkene donates into a Ag + empty metal orbital and back-donation occurs from a filled metal orbital into the alkene π* MO. Here I play a little "what if" game with this scenario to see what one can learn from doing so. Firstly, I will use
Identifiers
- UUID
- 82105f16-31de-4cee-a0b1-510b1df30b8e
- GUID
- http://www.ch.imperial.ac.uk/rzepa/blog/?p=10498
- URL
- https://www.ch.imperial.ac.uk/rzepa/blog/?p=10498
Dates
- Issued
-
2013-05-15T21:01:40
- Updated
-
2014-01-17T08:46:06
References
- Rzepa, H. S. (2013). Gaussian Job Archive for C4H4Pt(2+) [Data set]. figshare. https://doi.org/10.6084/m9.figshare.703546
- Rzepa, H. S. (2013). Gaussian Job Archive for C4H4Au(1+) [Data set]. figshare. https://doi.org/10.6084/m9.figshare.703547
- Rzepa, H. S. (2013). Gaussian Job Archive for C4H4Au(1+) [Data set]. figshare. https://doi.org/10.6084/m9.figshare.703576
- Rzepa, H. S. (2013). Gaussian Job Archive for C4H4Au(1+) [Data set]. figshare. https://doi.org/10.6084/m9.figshare.703577
