Helically conjugated molecules. A follow-up to [144]-annulene.
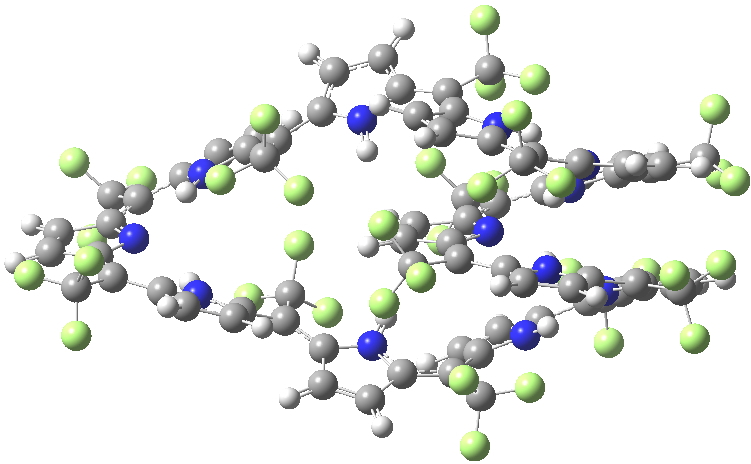
An extensive discussion developed regarding my post on a fascinating helical [144]-annulene. Topics included the nature of the ring current sustained by the π-electrons and in particular the bond-length alternation around the periphery and whether this should alter if the electron count were to be changed to that of a 4n+2 system (i.e. a dication). Whilst the [144]-annulene itself is hypothetical, it emerged that some compounds known as expanded porphyrins have very similar (albeit smaller scale) helical structures. X-ray structures for two such provide useful reality checks on the calculations. Here^‡ ^I include the (3D) coordinates of these two systems so that you can explore for yourself their helicity.

SELQUW. Click for 3D X-ray structure
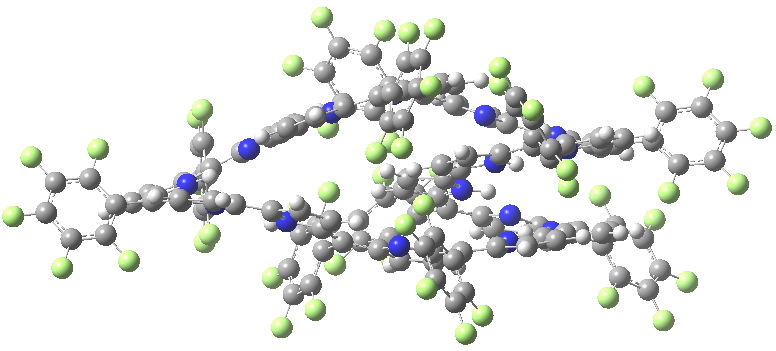
HIYTAL. Click for 3D X-ray structure
I include below Δrmeso, being the mean unsigned difference in bond length (Å) at the meso positions of the porphrin ring, the calculations being at the 6-311G(d,p) level using the DFT procedure indicated below. The linking number analysis[cite]10.1021/ja710438j[/cite] for such systems will be reported elsewhere.[cite]10.1021/ol703129z[/cite]
| Method | SELQUW | HIYTAL |
| X-ray | 0.048 | 0.045 |
| B97D/6-311G(d,p) | 0.025 | 0.015 |
| B3LYP/6-311G(d,p) | 0.047 | 0.017 |
‡The WordPress system operated here does not enable 3D coordinates to be inserted into the comment section of a post, only the main body.
Additional details
Description
An extensive discussion developed regarding my post on a fascinating helical [144]-annulene.
Identifiers
- UUID
- a6da8482-d348-49ad-8e13-b41014dba5ba
- GUID
- http://www.ch.imperial.ac.uk/rzepa/blog/?p=9512
- URL
- https://www.ch.imperial.ac.uk/rzepa/blog/?p=9512
Dates
- Issued
-
2013-02-12T12:41:08
- Updated
-
2014-01-17T08:32:57
References
- Rappaport, S. M., & Rzepa, H. S. (2008). Intrinsically Chiral Aromaticity. Rules Incorporating Linking Number, Twist, and Writhe for Higher-Twist Möbius Annulenes. Journal of the American Chemical Society, 130(24), 7613–7619. https://doi.org/10.1021/ja710438j
- Rzepa, H. S. (2008). Lemniscular Hexaphyrins as Examples of Aromatic and Antiaromatic Double-Twist Möbius Molecules. Organic Letters, 10(5), 949–952. https://doi.org/10.1021/ol703129z
