The Swern oxidation[cite]10.1016/0040-4020(78)80197-5[/cite] is a class of “activated” dimethyl sulfoxide (DMSO) reaction in which the active species is a chlorodimethylsulfonium chloride salt.
Rogue Scholar Beiträge
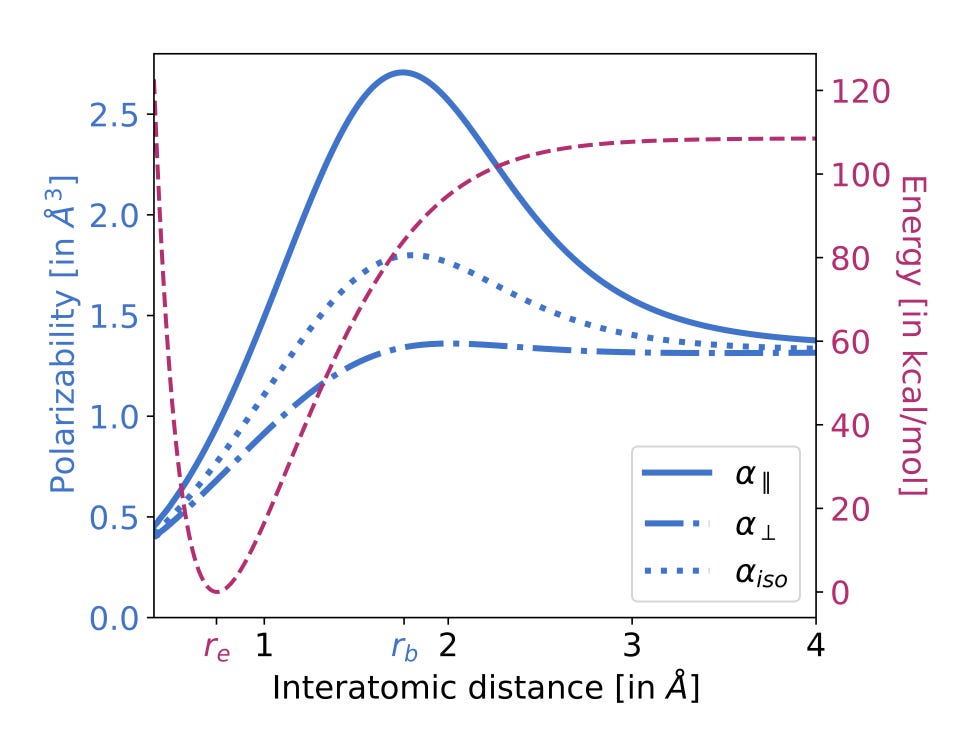
The Pauling model for enzymatic catalysis states that enzymes are “antibodies for the transition state”—in other words, they preferentially bind to the transition state of a given reaction, rather than the reactants or products. This binding interaction stabilizes the TS, thus lowering its energy and accelerating the reaction.
This is my last post on blogger.com. At least, that is the plan. It has been a great 18 years. I like to thank the owners of blogger.com and Google later for providing this service. I am continuing the chem-bla-ics on a new domain: https://chem-bla-ics.linkedchemistry.info/ I, like so many others, struggle with choosing open infrastructure versus the freebie model.
Some days ago, I started added boiling points to Wikidata, referenced from Basic Laboratory and Industrial Chemicals (wikidata:Q22236188), David R. Lide’s ‘a CRC quick reference handbook’ from 1993 (well, the edition I have). But Wikidata wants pressure (wikidata:P2077) info at which the boiling point (wikidata:P2102) was measured. Rightfully so. But I had not added those yet, because it slows me and can be automated with QuickStatements.
Just a quick note: I just love the level of detail Wikidata allows us to use. One of the marvels is the practices of named as, which can be used in statements for subject and objects. The notion and importance here is that things are referred to in different ways, and these properties allows us to link the interpretation with the source.

Respiratory pigments are metalloproteins that transport O 2 , the best known being the bright red/crimson coloured hemoglobin in human blood. The colour derives from Fe 2+ at the core of a tetraporphyrin ring. But less well known is blue blood , and here the colour derives from an oxyhemocyanin unit based on Cu 1+ (the de-oxy form is colourless) rather than iron.

Since the ostensible purpose of organic methodology is to develop reactions that are useful in the real world, the utility of a method is in large part dictated by the accessibility of the starting materials.
TW: stereotypes about molecular dynamics. In his fantastic essay “The Two Cultures,” C. P. Snow observed that there was (in 1950s England) a growing divide between the academic cultures of science and the humanities: He reflects on the origins of this phenomenon, which he contends is new to the 20th century, and argues that it ought to be opposed: Snow’s essay is wonderful: his portrait of a vanishing cultural intellectual unity

An important problem with simulating chemical reactions is that reactions generally take place in solvent, but most simulations are run without solvent molecules. This is a big deal, since much of the inaccuracy associated with simulation actually stems from poor treatment of solvation: when gas phase experimental data is compared to computations, the results are often quite good.
TW: sarcasm. Today, most research is done by academic labs funded mainly by the government. Many articles have been written on the shortcomings with academic research: Sam Rodriques recently had a nice post about how academia is ultimately an educational institution, and how this limits the quality of academic research. (It’s worth a read;