Continuing an exploration of the mechanism of this reaction, an alternative new mechanism was suggested in 1989 (having been first submitted to the journal ten years earlier!).[cite]10.1002/jhet.5570260518[/cite] Here the key intermediate proposed is a thiirenium cation (labelled 8 in the article) and labelled Int3 below.
The Willgerodt reaction[cite]10.1002/cber.18870200278[/cite], discovered in 1887 and shown below, represents a transformation with a once famously obscure mechanism. A major step in the elucidation of that mechanism came[cite]10.1021/ja01157a034[/cite] using the then new technique of 14 C radio-labelling, shortly after the atom bomb projects during WWII made 14 CO 2 readily available to researchers.
One of the most fascinating and important articles dealing with curly arrows I have seen is that by Klein and Knizia on the topic of C-H bond activations using an iron catalyst.[cite]10.1002/anie.201805511[/cite] These are so-called high spin systems with unpaired electrons and the mechanism of C-H activation involves both double headed (two electron) and fish-hook (single electron) movement.
Earlier, I explored the choreography or “timing”, of what might be described as the curly arrows for a typical taught reaction mechanism, the 1,4-addition of a nucleophile to an unsaturated carbonyl compound (scheme 1). I am now going to explore the consequences of changing one of the actors by adding the nucleophile to an unsaturated imine rather than carbonyl compound (scheme 2).
A little more than a year ago, a ChemRxiv pre-print appeared bearing the title referenced in this post,[cite]10.26434/chemrxiv.8009633.v1[/cite] which immediately piqued my curiosity. The report presented persuasive evidence, in the form of trapping experiments, that dicarbon or C 2 had been formed by the following chemical synthesis.
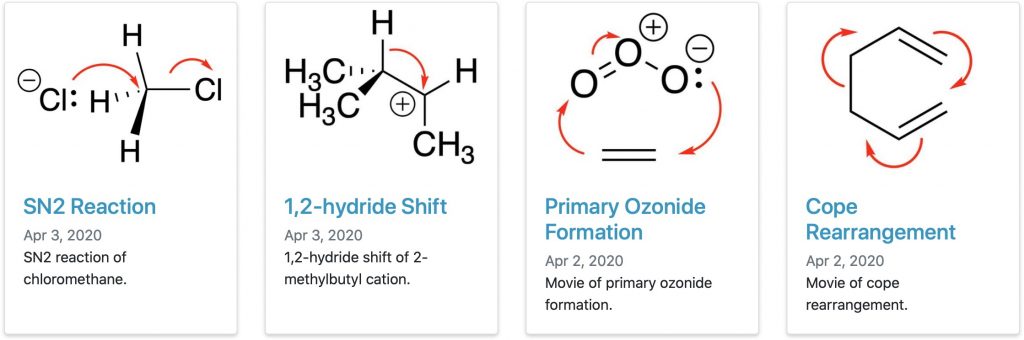
In a previous post, I talked about a library of reaction pathway intrinsic reaction coordinates (IRCs) containing 115 examples of organic and organometallic reactions. Now (thanks Dean!) I have been alerted to a brand new databank of dynamics trajectories (DDT), with the focus on those reactions taught in undergraduate organic chemistry courses, some of which are shown below.
A reaction can be thought of as molecular dancers performing moves. A choreographer is needed to organise the performance into the ballet that is a reaction mechanism. Here I explore another facet of the Michael addition of a nucleophile to a conjugated carbonyl compound.
In the previous post, I looked at the mechanism for 1,4-nucleophilic addition to an activated alkene (the Michael reaction). The model nucleophile was malonaldehyde after deprotonation and the model electrophile was acrolein (prop-2-enal), with the rate determining transition state being carbon-carbon bond formation between the two, accompanied by proton transfer to the oxygen of the acrolein.
In 2013, I created an iTunesU library of 115 mechanistic types in organic and organometallic chemistry, illustrated using video animations of the intrinsic reaction coordinate (IRC) computed using a high level quantum mechanical procedure. Many of those examples first derived from posts here. That collection is still available and is viewable in the iTunesU app on an iPhone or an iPad.
If, as a synthetic chemist, you want to invert the configuration of an alcohol in which the OH group is at a chiral centre, then the Mitsunobu reaction has been a stalwart for many years. Now a catalytic version has been published, [cite]10.1126/science.aax3353[/cite] along with a proposed mechanism.
