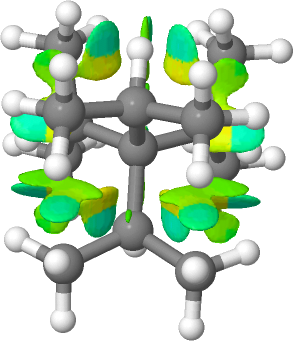
The homologous hydrocarbon series R 4 C is known for R=Me as neopentane and for R=Et as 3,3-diethylpentane.

The homologous hydrocarbon series R 4 C is known for R=Me as neopentane and for R=Et as 3,3-diethylpentane.
Whilst I was discussing the future of scientific publication in the last post, a debate was happening behind the scenes regarding the small molecule cyclopropenylidene. This is the smallest known molecule displaying π-aromaticity, but its high reactivity means that it is unlikely to be isolated in the condensed phase.
Occasionally, someone comments about an old post here, asking a question. Such was the case here, when a question about the dipole moment of cyclopropenylidene arose. It turned out to be 3.5D, but this question sparked a thought about the related molecule below.
From the last few posts here, you might have noticed much discussion about how the element carbon might sustain a quadruple bond. The original post on this topic from some years ago showed the molecular orbitals of the species CN + , which included two bonding π-types and a low lying nodeless bonding σ-orbital, all with double occupancies and adding up to a triple bond.
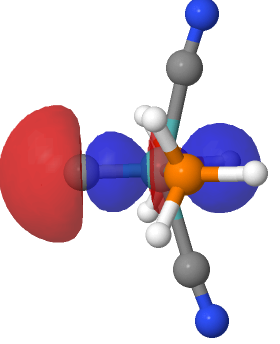
I noted in an earlier post the hypothesized example of (CO) 3 Fe⩸C[cite]10.1039/d0cp03436c[/cite] as exhibiting a carbon to iron quadruple bond and which might have precedent in known five-coordinate metal complexes where one of the ligands is a “carbide” or C ligand. I had previously mooted that the Fe⩸C combination might be replaceable by an isoelectronic Mn⩸N pair which could contain a quadruple bond to the nitrogen.
The proposed identification of molecules with potential metal to carbon quadruple bonds, in which the metal exhibits trigonal bipyramidal coordination rather than the tetrahedral modes which have been proposed in the literature[cite]10.1039/d0cp03436c[/cite],[cite]10.1039/d1cp00598g[/cite],[cite]10.1021/acs.jpclett.9b03484[/cite] leads on to asking whether simple trigonal coordination at the metal can also sustain this theme?
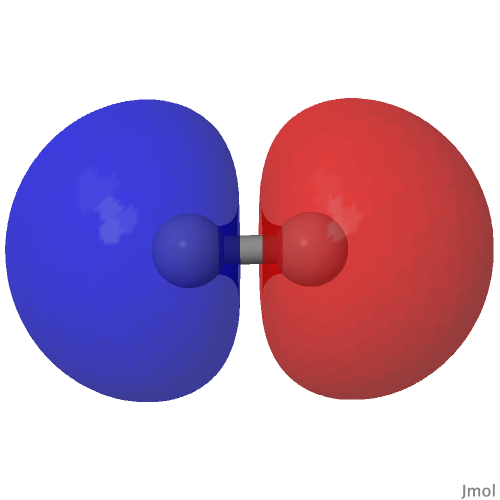
Introductory chemistry will tell us that a triple bond between say two carbon atoms comprises just one bond of σ-axial symmetry and two of π-symmetry. Increasingly mentioned nowadays is the possibility of a quadruple bond between carbon and either itself or a transition metal, as discussed in the previous post. Such a bond comprises TWO bonds of σ-axial symmetry.
Following from much discussion over the last decade about the nature of C 2 , a diatomic molecule which some have suggested sustains a quadruple bond between the two carbon atoms, new ideas are now appearing for molecules in which such a bond may also exist between carbon and a transition metal atom.
Deltamethin is a pyrethroid insecticide for control of malaria which has been used for a little while. Perhaps inevitably, mosquitoes are developing resistance to it. So what could be done about countering this? Well, perhaps surprisingly, form a polymorph![cite]10.1073/pnas.2013390117[/cite] These crystal structure isomers are often highly undesirable;
For obvious reasons, anti-viral molecules are very much in the news at the moment. Thus Derek Lowe highlights Molnupiravir which is shown as a hydroxylamine, the representation originating from the Wikipedia page on the molecule. I like stereocentres more clearly identified using eg R / S notation and so I went to another source of information, SciFinder, which represents the molecule in a different way.