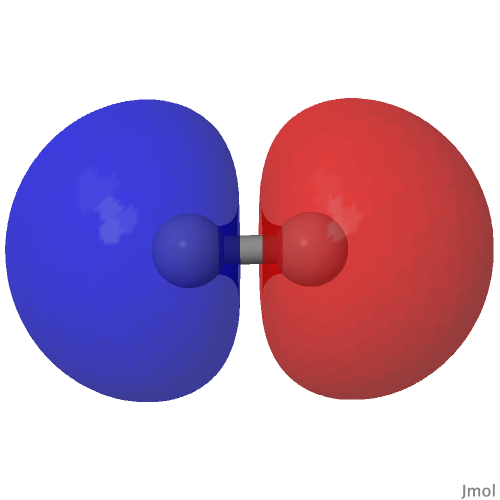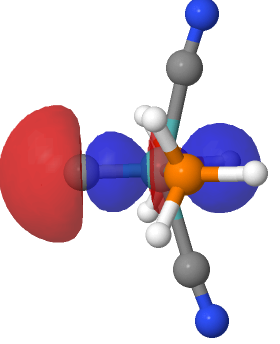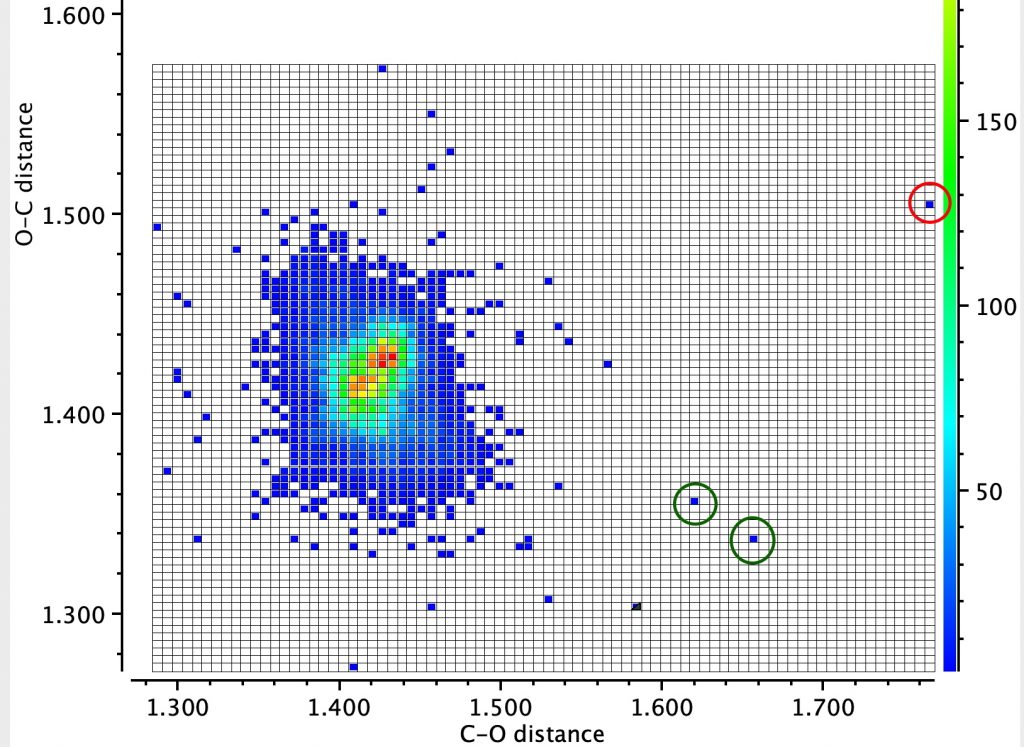
In the earlier post on the topic of anomeric effects, I identified a number of outliers associated with large differences in the lengths of two carbon-oxygen bonds sharing a common carbon atom. Here is another of these outliers (MUZZIS[cite]10.1107/S2056989016002899[/cite]) which shows equally unusual properties.