
It is a sign of the times that one travels to a conference well-connected. By which I mean email is on a constant drip-feed, with venue organisers ensuring each delegate receives their WiFi password even before their room key.

It is a sign of the times that one travels to a conference well-connected. By which I mean email is on a constant drip-feed, with venue organisers ensuring each delegate receives their WiFi password even before their room key.

This is taking place in the idyllic surroundings of the Niederwald forest, Rüdesheim, Germany. Here I highlight only aspects of the first three talks.
Mention carbon dioxide (CO 2 ) to most chemists and its properties as a metal ligand are not the first aspect that springs to mind. Here thought I might take a look at how it might act as such.
Research data (and its management) is rapidly emerging as a focal point for the development of research dissemination practices.
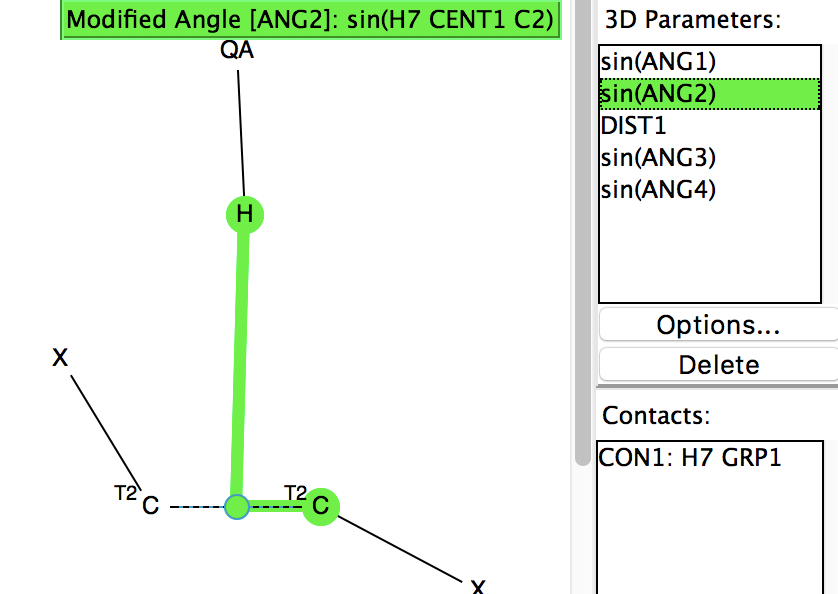
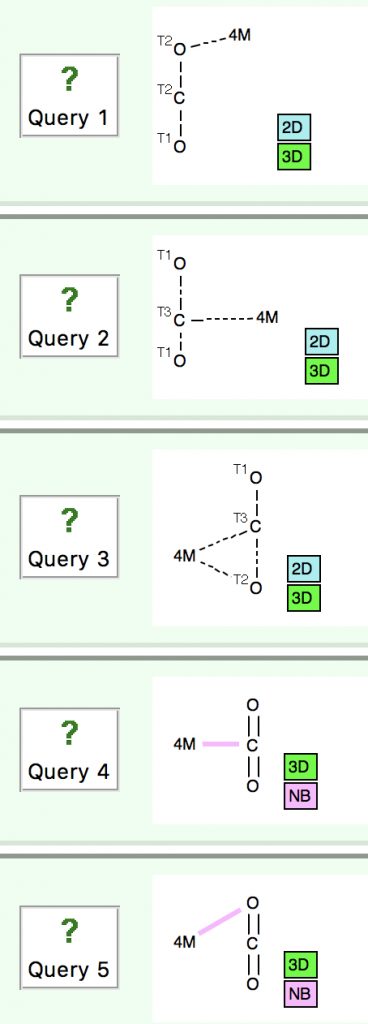
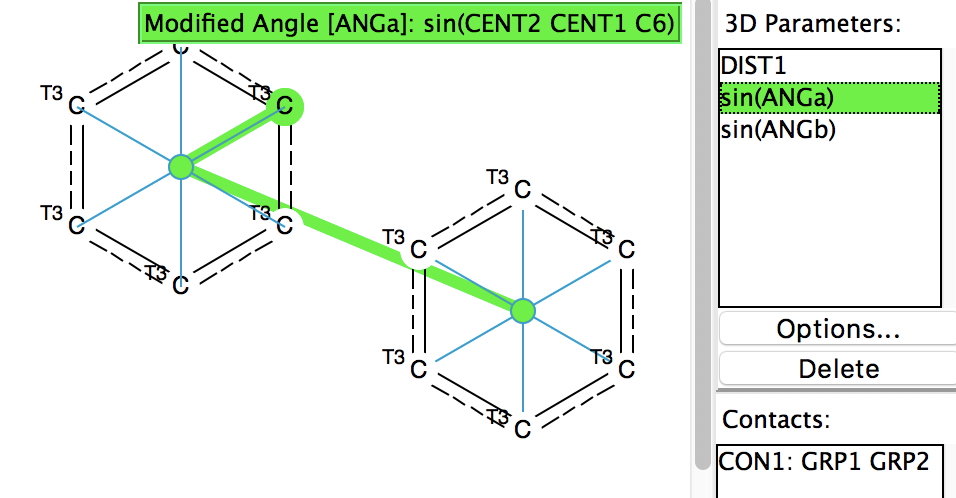
Following on from my re-investigation of close hydrogen bonding contacts to the π-face of alkenes, here now is an updated scan for H-bonds to alkynes. The search query (dataDOI: 10.14469/hpc/2478) is similar to the previous one: QA is any of N,O,F,Cl. X is any atom, including metals and non-metals.
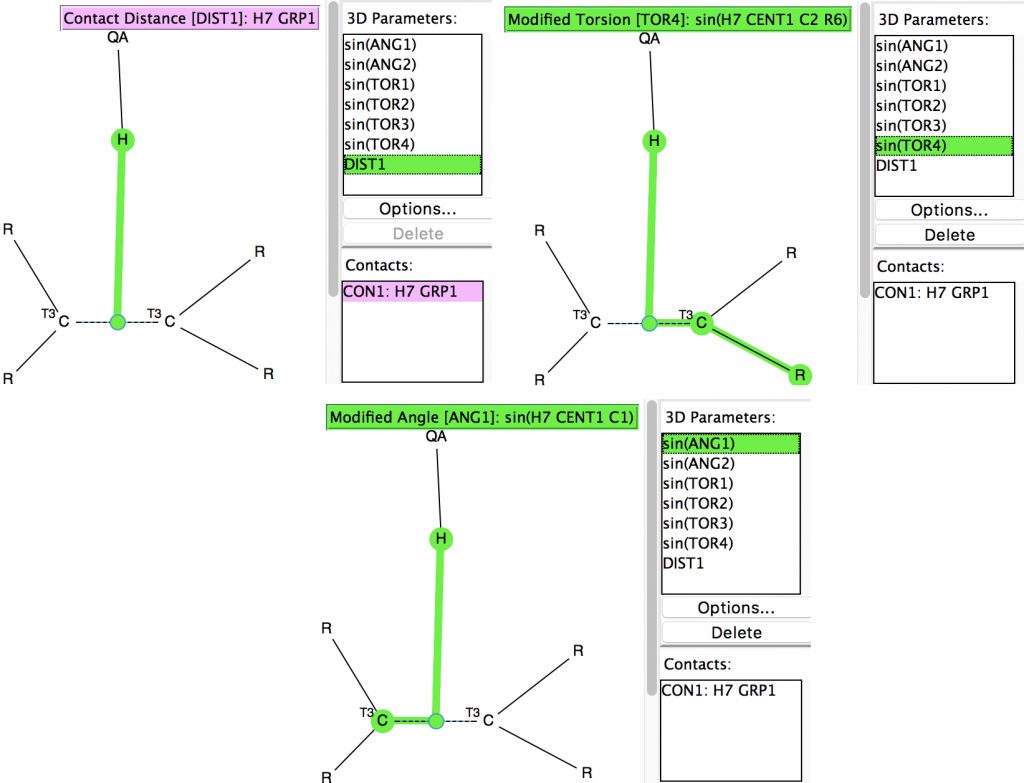
Back in the early 1990s, we first discovered the delights of searching crystal structures for unusual bonding features.[cite]10.1039/P29940000703[/cite] One of the first cases was a search for hydrogen bonds formed to the π-faces of alkenes and alkynes. In those days the CSD database of crystal structures was a lot smaller (<80,000 structures; it’s now ten times larger) and the search software less powerful.
Layer stacking in structures such as graphite is well-studied. The separation between the π-π planes is ~3.35Å, which is close to twice the estimated van der Waals (vdW) radius of carbon (1.7Å). But how much closer could such layers get, given that many other types of relatively weak interaction such as hydrogen bonding can contract the vdW distance sum by up to ~0.8Å or even more?
Following my conformational exploration of enols, here is one about a much more common molecule, a carboxylic acid. The components of the search are shown as four queries below, which will be combined in various Boolean senses (DOI: 10.14469/hpc/2462). Query one defines the carboxylic acid, with 3-coordinate carbon specified at the carbonyl along with 1-coordinate for the carbonyl oxygen.
Both the cyclopropenium cation and the cyclopentadienide anion are well-known 4n+2-type aromatic ions, but could the two together form an ion-pair? A search of the Cambridge structure database reveals 52 instances of the cyclopropenium cation with a variety of counter-anions, 77 cyclopentadienide anions with a variety of counter-cations and one (SOWMOG, private communication to CSD) where the two sub-structures are common.
Enols are simple compounds with an OH group as a substituent on a C=C double bond and with a very distinct conformational preference for the OH group. Here I take a look at this preference as revealed by crystal structures, with the theoretical explanation.