This second report highlights two “themes”, or common ideas that seem to emerge spontaneously from diversely different talks. Most conferences do have them. The first is “ embedding ”, which in this context means treating different parts of a probably complex molecular system at different levels of theory.
Postagens de Rogue Scholar

I am attending a conference. Plenaries at such events can sometimes provide interesting pointers on things to come (and sometimes they simply point to things past). At WATOC2014 in Santiago Chile, the first plenary was by Paul Ayers with the impressive title “ Concepts for organising chemical knowledge ” which certainly sounds as if it is pointing forward!
The outcome of pericyclic reactions con depend most simply on three conditions, any two of which determine the third. Whether the catalyst is Δ or hν (heat or light), the topology determining any stereochemistry and the participating electron count (4n+2/4n). It is always neat to conjure up a simple switch to toggle these; heat or light is simple, but what are the options for toggling the electron count?
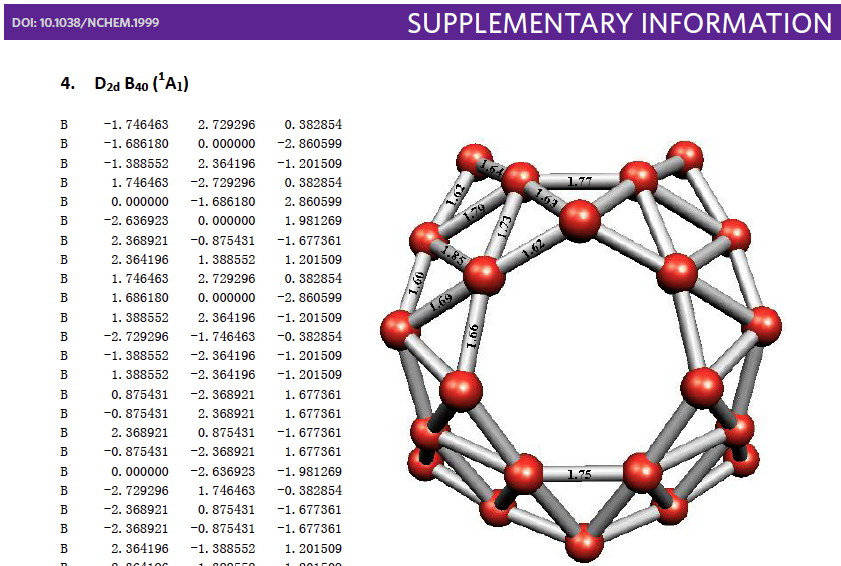
Whilst clusters of carbon atoms are well-known, my eye was caught by a recent article describing the detection of a cluster of boron atoms, B 40 to be specific.[cite]10.1038/nchem.1999[/cite] My interest was in how the σ and π-electrons were partitioned. In a C 40 , one can reliably predict that each carbon would contribute precisely one π-electron. But boron, being more electropositive, does not always play like that.
The Bürgi–Dunitz angle describes the trajectory of an approaching nucleophile towards the carbon atom of a carbonyl group. A colleague recently came to my office to ask about the inverse, that is what angle would an electrophile approach (an amide)? Thus it might approach either syn or anti with respect to the nitrogen, which is a feature not found with nucleophilic attack.
Following the discussion here of Kekulé’s suggestion of what we now call a vibrational mode (and which in fact now bears his name), I thought I might apply the concept to a recent molecule known as [2.2]paracyclophane.
Ribulose-1,5-bisphosphate reacts with carbon dioxide to produce 3-keto-2-carboxyarabinitol 1,5-bisphosphate as the first step in the biochemical process of carbon fixation.

I have just noticed unexpected links between two old posts, one about benzene, one about diphenyl magnesium and a link to August Kekulé. † Click for 3D The post about benzene dealt with the apparently simple issue of why all the C-C bonds are of equal length. The answer, in brief is purely because of the σ-electrons. If the π-electrons had their way, benzene would have three short and three long C-C bonds.
The journal of chemical education can be a fertile source of ideas for undergraduate student experiments. Take this procedure for asymmetric epoxidation of an alkene.[cite]10.1021/ed077p271[/cite] When I first spotted it, I thought not only would it be interesting to do in the lab, but could be extended by incorporating some modern computational aspects as well.
Around 100 tons of the potent antimalarial artemisinin is produced annually; a remarkable quantity given its very unusual and fragile looking molecular structure (below). When I looked at this, I was immediately struck by a thought: surely this is a classic molecule for analyzing stereoelectronic effects (anomeric and gauche). Here this aspect is explored.