The ultimate reduction in size for an engineer is to a single molecule. It’s been done for a car; now it has been reported for the pixel (picture-element).[cite]10.1021/ja404256s[/cite] The molecule above (X=O, NR, R=aryl, etc) has been shown to be capable of acting as a molecular pixel.
Postagens de Rogue Scholar
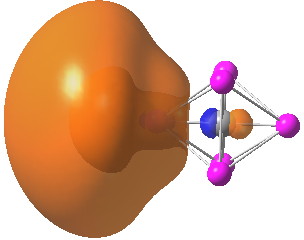
A comment made on the previous post on the topic of hexa-coordinate carbon cited an article entitled “ Observation of hypervalent CLi 6 by Knudsen-effusion mass spectrometry ”[cite]10.1038/355432a0[/cite] by Kudo as a amongst the earliest of evidence that such species can exist (in the gas phase). It was a spectacular vindication of the earlier theoretical
Sometimes the originators of seminal theories in chemistry write a personal and anecdotal account of their work. Niels Bohr[cite]10.1007/BF01326955[/cite] was one such and four decades later Robert Woodward wrote “ The conservation of orbital symmetry ” (Chem. Soc. Special Publications (Aromaticity), 1967 , 21 , 217-249;
A few posts back, I explored the “benzidine rearrangement” of diphenyl hydrazine. This reaction requires diprotonation to proceed readily, but we then discovered that replacing one NH by an O as in N,O-diphenyl hydroxylamine required only monoprotonation to undergo an equivalent facile rearrangement. So replacing both NHs by O to form diphenyl peroxide (Ph-O-O-Ph) completes this homologous series.
I recently got an email from a student asking about the best way of rationalising epoxide ring opening using some form of molecule orbitals.
My previous dissection of the mechanism for ester hydrolysis dealt with the acyl-oxygen cleavage route (red bond). There is a much rarer[cite]10.1039/jr9550001522[/cite] alternative: alkyl-oxygen cleavage (green bond) which I now place under the microscope.
The mechanism of ester hydrolysis is a staple of examination questions in organic chemistry. To get a good grade, one might have to reproduce something like the below. Here, I subject that answer to a reality check.
The electrophilic substitution of indoles is a staple of any course on organic chemistry.
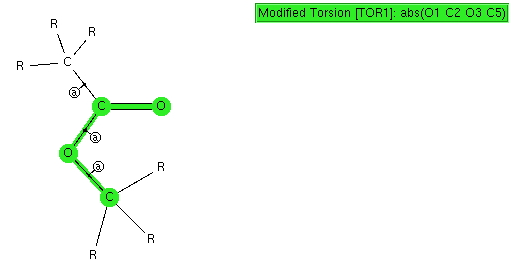
I conclude my exploration of conformational preferences by taking a look at esters. As before, I start with a search definition, the ester being restricted to one bearing only sp 3 carbon centers. The result of such a search is pretty clear-cut; they all exist in just one conformation, the s-cis , in which a lone pair of electrons on the alkyl-oxygen is aligned quite precisely anti-periplanar with the axis of the C=O bond.
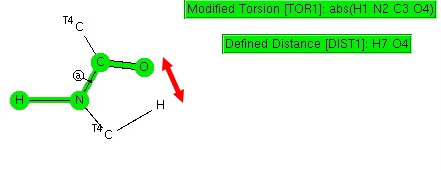
Amides with an H-N group are a component of the peptide linkage (O=C-NH). Here I ask what the conformation (it could also be called a configuration) about the C-N bond is. A search of the following type can be defined: The dihedral shown is for H-N-C=O (but this is equivalent to the C-C-N-C dihedral, which is also often called the dihedral angle associated with the peptide group). I have also added a distance, from a C-H to the