Hypervalency: Is it real?

The Wikipedia page on hypervalent compounds reveals that the concept is almost as old as that of normally valent compounds. The definition there, is “a molecule that contains one or more main group elements formally bearing more than eight electrons in their valence shells” (although it could equally apply to e.g. transition elements that would contain e.g. more than 18 electrons in their valence shell). The most extreme example would perhaps be of iodine (or perhaps xenon). The normal valency of iodine is one (to formally complete the octet in the valence shell) but of course compounds such as IF7 imply the valency might reach 7 (and by implication that the octet of electrons expands to 14). So what of IF7? Well, there is a problem due to the high electronegativity of the fluorine. One could argue that the bonds in this molecule are ionic, and hence that the valence electrons really reside in lone pairs on the F. Thus the apparently hypervalent PF5 could be written PF4+…F–, in which case the P is not really hypervalent after all. We need a compound with un-arguably covalent bonds. Well, what about IH7? One might probably still argue about ionicity (for example H+…IH6–) but that puts electrons on I and not H, and hence does not change any hypervalency on the iodine. Surely, if hypervalency is a real phenomenon, it should manifest in IH7?

IH7. A true hypervalent molecule? Click for 3D
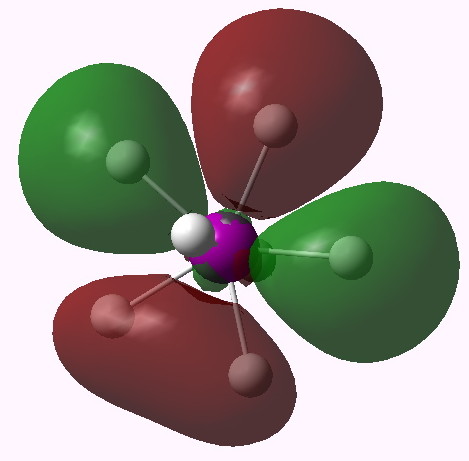 MO 16 (E2'). Click for 3D |
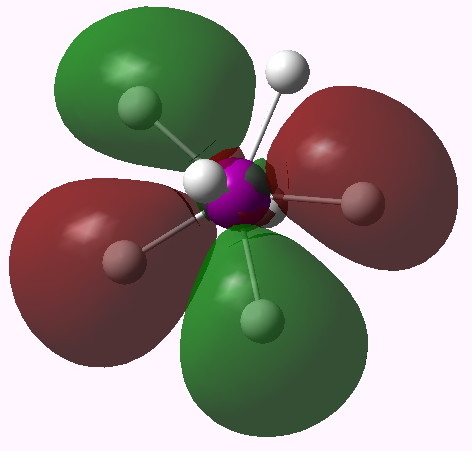 MO 15 (E2') |
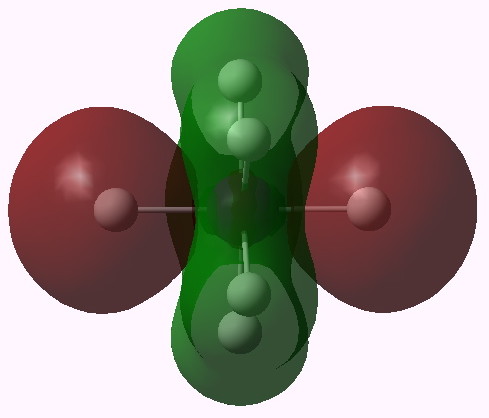 MO 14 (A1') |
|
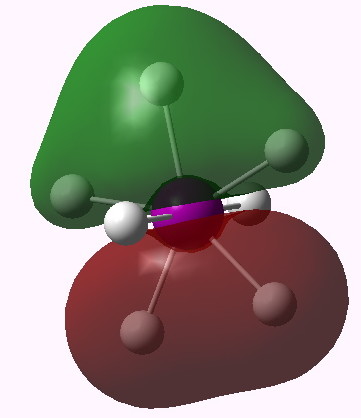 MO 13 (E1') |
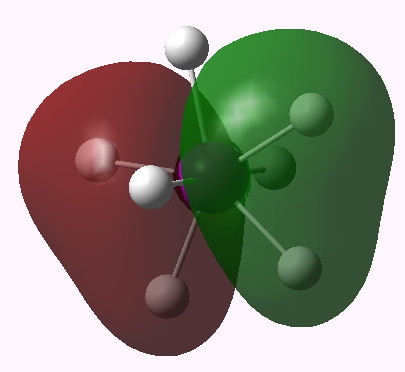 MO 12 (E1') |
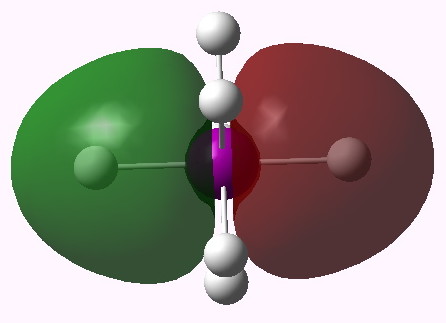 MO 11 (A2'') |
|
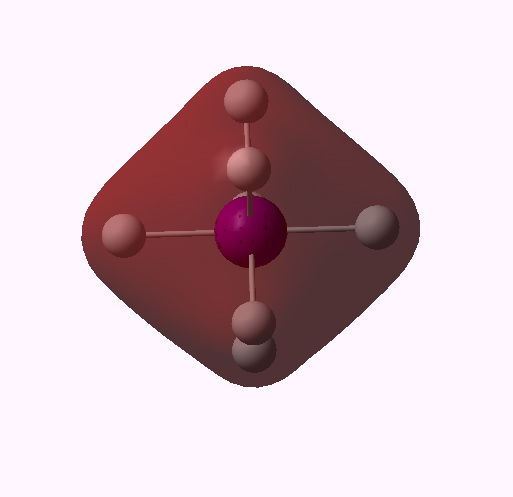 MO 10 (A1') |
|
One notices that whilst orbitals 10-14 are clearly bonding in the I-H region, orbitals 15-16 seem antibonding in that region (there is a node along the I-H bond). We are seeing much the same phenomenon that occurs when the bond order of 3 in N2 is reduced to 1 in F2 due to occupancy of anti-bonding orbitals. Can this be quantified? NBO (5.9) analysis reveals the following.
- The effective (pseudopotential) core has 28 electrons, and the outer core 18. The valence orbitals contain 13.79, and only 0.21 electrons are Rydberg (higher shell). So little occupancy of e.g. 6s/6p then!
- The Wiberg bond index indicates each H has a total bond order of very close to 1 (its natural valence state), whilst I is 3.45. Remember that the maximum total bond index of a covalently bound atom using a pure octet of valence electrons is 4 (8/2, think carbon). The iodine is NOT hypervalent!
- So why, if its not hypervalent, is it so strongly hypercoordinate? Well, there are 14 valence electrons, but they do NOT all occupy the I-H regions, which have bond orders between 0.46 (equatorial) or 0.57 (axial). The only other place they can be is in the H-H regions! Consider the bond order values between say hydrogen 2 to the four other equatorial atoms 4,5,7,8. They are respectively 0.05, 0.15, 0.05, 0.15. So each hydrogen has a total bond order of 1, but only slightly more than half of this comes from the I-H, the rest comes from H-H. To put it another (approximate) way, of the 14 valence electrons, ~8 might be considered to be associated with I-H bonds, and ~6 with H-H bonds. The hypervalency has been in effect absorbed into the H-H regions. This means no atom in this molecule is at all hypervalent.
- Well, if the iodine is not hypervalent, and some of its valence electrons occupy I-H anti-bonding orbitals, why is it stable at all (in the sense that all the vibrations are real, and it’s clearly a minimum in the potential energy surface). Here, I merely speculate. Iodine has a large core charge, and hence the inner core electrons are starting to exhibit relativistic contractions. This effect stabilizes the outer 5s/5p electrons, and so occupancy of anti-bonding MOs generated from such AOs is not so unfavourable as one might expect. Perhaps, the hypercoordination shown by IH7 is after all a relativistic effect rather than a hypervalent effect? In which case, what will AsH7 show?
So, what might have been an archetypal covalent hypervalent molecule is no such! IH7 shows entirely normal valencies, one for H and the iodine does not even reach four (if anything, its sub-valent rather than hypervalent). One may be entitled to ask if ANY main group element exhibits hypervalency!
Additional details
Description
The Wikipedia page on hypervalent compounds reveals that the concept is almost as old as that of normally valent compounds.
Identifiers
- UUID
- 3ef2551b-b06d-484a-8aaa-202f42584b0d
- GUID
- http://www.ch.ic.ac.uk/rzepa/blog/?p=2599
- URL
- https://www.ch.ic.ac.uk/rzepa/blog/?p=2599
Dates
- Issued
-
2010-10-16T13:10:21
- Updated
-
2010-10-18T15:40:19
